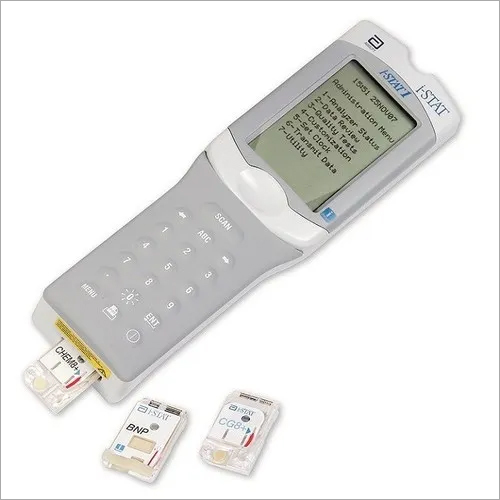
The need to construct another treatment area has been consequently avoided, providing the hospital a significant amount of saved cost. It also saved them about €62,208 worth of staff cost per year for troponin tests alone. The installing of i-STAT greatly reduced the previous daily peak of 40 patients to 33 patients. Aside from the lengthened stay, this also meant increased waiting times for patients, stressful working situations, and higher costs caused by additional staff and overtime payments. 
On Octothe firm distributed Urgent Medical Device Recall Communication letters by mail. The observed rate of quality check codes could be in the 3 to 5 range. Before i-STAT, it took Augsburg Hospital’s Central Lab around 42 to 121 minutes to perform a troponin test. i-STAT¿ cTnI blue cartridges may generate a higher than expected number of quality check codes. Any printed copies are uncontrolled unless specified. With majority of the patients suffering from non-specific chest pains, troponin tests are in high demand. Medical Approval: Dr Benjamin Jung Medical Approval Date: Version: 1.2 Implementation Date: 12:22:44 AM Folder Name: CW\Point of Care\Blood Gas Electrolytes - iSTAT This is a controlled document for CW use only. Length of stay is a considerable factor in crowd control. This may be due to the growing numbers in the elderly population in Europe. Recent reports observed that their ED visits increased by at least 7% to 10%. With an average of 200 patients coming in daily (roughly 70,000 visitors per year), the staff had to constantly face their limitations in man power, treatment area, and equipment. You can download a raw copy of the database here.For Augsburg Hospital’s ER department, overcrowding is a rising concern. Always cite the International Consortium of Investigative Journalists when using this data. The International Medical Devices Database is licensed under the Open Database License and its contents under Creative Commons Attribution-ShareAlike license. Context: Point-of-care testing (POCT) for blood gas and chemistries is widely adopted in the emergency department (ED) for fast turnaround and critical medical. This database is not intended to provide medical advice and patients should check with their doctors to determine if it contains relevant information and if such information has medical implications for them. The same device may have different names in different countries. We are not suggesting or implying that any companies or other entities included in the International Medical Devices Database engaged in unlawful conduct or otherwise acted improperly. i-stat act is similar to traditional act tests except that the endpoint is indicated by a chemical measurement of the presence of thrombin instead of a mechanical measurement of a physical clot. 
Controls can be stored at room temperature (20 to 300C) for 5 days. These solutions do not contain human serum or serum products. Medical devices help to diagnose, prevent and treat many injuries and diseases. with i-stat act, healthcare professionals can measure the time required for complete activation of the coagulation cascade. Each vial contains 5 ml of an aqueous buffered solution with preservatives, electrolytes, glucose and urea equilibrated with precise mixtures of O2, CO2, and N2 gases. Explore more than 120,000 Recalls, Safety Alerts and Field Safety Notices of medical devices and their connections with their manufacturers.ĭo you work in the medical industry? Or have experience with a medical device? Our reporting is not done yet.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
